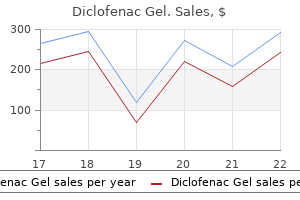
Diclofenac gel 20 gm generic amexExamples of polyacrylic acid-based polymers are carbopol arthritis in back hereditary diclofenac gel 20 gm buy line, polycarbophil arthritis in the fingers symptoms cheap diclofenac gel 20 gm online, polyacrylic acid, polyacrylate, poly(methylvinylether-co-methacrylic acid), poly(2-hydroxyethyl methacrylate), and poly(methacrylate). In the case of local drug delivery, similar to vaginal drug supply, the utilization of mucoadhesive polymer in the drug delivery system can lead to higher residence time and prolonged length of native motion of the medicine. The first contact of the mucoadhesive polymer with the mucosal surface leads to surface adhesion because of a number of favorable hydrogen-bond and electrostatic interactions and polymer growth due to water uptake and plasticization of the drug supply system. The polymer and the drug supply system increase and spread on the mucosal floor. The subsequent robust bonding (adhesion) between the polymer and the mucus is a function of polymer chain diffusion, hydration and plasticization, and interlocking bond formation. Attractive interactions between the hydrophilic mucoadhesive polymer and the hydrophilic polymeric glycoproteins within the mucus lead to mutual entanglement and interpenetration of the polymeric chains. This facilitates the formation of extra and deeper electrostatic and hydrogen-bond interactions, which promote bioadhesion or mucoadhesion. Thus, mucoadhesion is facilitated by the presence of hydrogen-bond-forming groups within the polymeric chain, flexibility of the polymer chains, and the surface exercise of the drug supply system. Mechanical forces at the site of adhesion may help in deeper penetration and mechanical interaction of the polymers. Thus, Sm = F A 282 Pharmaceutical Dosage Forms and Drug Delivery the Sm could be calculated in vitro on the isolated mucus immobilized on a man-made floor or ex vivo, utilizing a organic surface, such as an isolated intestinal lumen. In vivo assessment of bioadhesion is normally done by measuring the residence time of the dosage form on the site of bioadhesion by an imaging technique. Buccal drug delivery systems search to ship drug domestically into the oral cavity for local treatment of oral lesions. When used to deliver a drug to the systemic circulation, corresponding to by sublingual administration, bypassing the hepatic first-pass metabolism can contribute to greater bioavailability. Similarly, drug delivery to the nasal mucosa and the vaginal tissue is utilized for local drug motion or rapid drug absorption in the systemic circulation, bypassing the hepatic first-pass metabolism. Ocular drug supply using mucoadhesive polymers seeks to tackle the issue of extreme drainage of the drug by way of the lachrymal glands earlier than enough absorption can take place. Prolonged retention of the drug on the cornea reduces precorneal drainage loss of the drug and increases the duration of drug absorption, thus enhancing ocular bioavailability. Mucoadhesive polymers adhere to the mucin coat overlaying the conjunctiva and the corneal floor of the eye. Ocular mucoadhesion markedly prolongs the residence time of a drug within the conjunctival sac, since clearance of a mucoadhesive dosage kind is controlled by the much slower fee of mucus turnover somewhat than the tear turnover fee. Incorporation of mucoadhesive polymers, such as chitosan, poly(acrylic acid), alginate, poly(methacrylic acid), and sodium carboxymethyl cellulose, into the oral solid drug delivery methods can enhance the residence time of the drug and adhesion of particulate drug to the mucosal floor, resulting in greater native concentration at the website of drug absorption. The selection of dosage type is decided by the route of drug administration as well as the desired traits of the drug delivery system. For instance, while tablet and granules are appropriate for administration by way of the oral route, options are more suitable for ocular and nasal drug supply, patches for transdermal drug supply, films for buccal drug delivery, and gels and ointments for vaginal drug supply. Percutaneous absorption of a drug usually results from direct penetration of the drug through the stratum corneum, deeper epidermal tissues, and the dermis. When the drug reaches the vascularized dermal layer, it becomes obtainable for absorption into the overall circulation. Among the factors influencing percutaneous absorption are the physicochemical properties of the drug, together with its molecular weight, solubility, partition coefficient, nature of vehicle, and situation of the pores and skin. Chemical permeation enhancers, iontophoresis, or both are sometimes used to improve the percutaneous absorption of a drug. In common, patches are composed of three key compartments: a protecting seal that varieties the exterior floor and protects it from harm, a compartment that holds the medication itself and has an adhesive backing to maintain the complete patch on the pores and skin floor, and a release liner that protects the adhesive layer during storage and is removed just before application. Most patches belong to one of the two common types-the reservoir system and the matrix system. The reservoir system incorporates the drug in a compartment of the patch, which is separated from the adhesion floor. Drug transport from the patch to the skin in channelized and managed by way of a rate-limiting floor layer. Diffusion of the drug via the polymer matrix and the bioadhesive properties of the polymer determine the rate of drug absorption. Marketed transdermal patches are exemplified by Estraderm (estradiol), Testoderm (testosterone), Alora (estradiol), Androderm (testosterone), and Transderm-Scop (scopolamine). The higher the diploma of crystallinity of the polymer, the decrease the speed of dissolution. Molecular weight and molecular weight distribution affect solvent penetration and crystallinity. Increase within the main-chain polarity will increase the glass transition temperature of a polymer. Modification of biomaterial surfaces with polyethylene glycol minimizes protein adsorption and/or platelet adhesion. What is the number common molecular weight (M n) and the weight common molecular weight (Mw) Katagiri K, Nakamura M, and Koumoto K (2010) Magnetoresponsive sensible capsules fashioned with polyelectrolytes, lipid bilayers and magnetic nanoparticles. Discuss pseudoplastic and dilatant rheograms and establish shearthinning and shear-thickening phenomena. The flow of easy liquids may be described by viscosity, an expression of the resistance to flow. Rheological properties are important considerations within the manufacturing, evaluation, and use of a quantity of dosage types, including solutions, emulsions, suspensions, pastes, lotions, suppositories, parenteral injectable drug merchandise, and intravenous infusions. Viscosity is necessary for the blending and circulate of supplies, their packaging into containers, and their removal earlier than use-whether achieved by pouring from a bottle, extrusion from a tube, or 287 288 Pharmaceutical Dosage Forms and Drug Delivery passage by way of a syringe needle. For example, pourability, spreadability, and syringeability of an emulsion are decided by its rheological properties. The constant that relates the circulate of a liquid to the applied stress known as viscosity. Viscosity can also be expressed as the constant that relates the shearing stress (force per unit area utilized parallel to the direction of circulate of liquid) and shear fee (volume of fluid move per unit area of the liquid). The more commonly used unit of viscosity is dyne*second/cm 2, which is named Poise (P). The higher the viscosity of a liquid, the higher the shearing stress required to produce a certain rate of shear. Increased Brownian movement, fast motion of liquid molecules amongst each other, at higher temperatures reduces power expense from the shearing drive in overcoming intermolecular cohesion forces, resulting in a higher proportion of energy utilized in influencing the path of flow. Thus, from the temperature of highest viscosity of the fluid, slight changes in temperature result in far more change in fluid viscosity in contrast with the impact of temperature on viscosity near the bottom viscosity of a fluid. The temperature�viscosity relationships are important for routine dealing with of liquids in pharmaceutical manufacturing under ambient temperature circumstances and handling of drug products by patients. For example, an injectable solution saved in a refrigerator may have a lot higher viscosity and could additionally be troublesome to inject than the identical answer brought to room temperature before injection. Plastic circulate is related to, for instance, the presence of flocculated particles in concentrated suspensions. Flocculated solids are gentle, fluffy conglomerates of adjoining particles held together by weak van der Waals forces.
Buy diclofenac gel 20 gm with visaIf the guide wire preferentially enters the descending aorta from the brachiocephalic trunk arthritis fat fingers buy diclofenac gel 20 gm with visa, a fastidiously timed inspiration is commonly effective in redirecting the advancing guide wire into the aortic root arthritis pain in feet buy generic diclofenac gel 20 gm line. Heparin is routinely administered to patients present process radial angiography to preserve radial artery patency. Coronary anatomy: normal variants Usual coronary anatomy consists of two coronary ostia, one in the left coronary sinus and the other in the best coronary sinus. Other variations embody ectopic origins of the coronary arteries and customary ostia. These are rare however can pose challenges throughout angiography, with particular reference to catheter choice. Rarely, aberrant coronary anatomy may be associated with an increased danger of sudden death or symptomatic ischaemia on account of exterior arterial compression by the good vessels. Both a small aneurysmal space (a) and filling defect (fd) are seen, indicating severe atherosclerotic illness. A Terumo wire was handed by way of the diseased area and the case accomplished using a long change wire. Different configurations and dimensions may be selected depending on the anatomy of the aortic root and site of the coronary ostia. Catheter engagement the coronary ostia are engaged by careful development and torque of the appropriate catheter and this can be a crucial moment during any coronary procedure. This disastrous complication is most probably to happen within the presence of pre-existing ostial disease A and may result from poorly managed ostial intubation, especially with a catheter which is malaligned or undersized. Subsequent high-pressure distinction injection into a catheter whose tip is angulated into the ostial vessel wall is doubtlessly catastrophic as an extending dissection flap can hinder luminal flow and may progress to aortic root dissection or perforation. This is usually instantly apparent by lack of arterial strain upon engagement. Subsequent distinction injection will reveal the extent of ostial disease with little backflow of distinction into the aortic root. Even a brief interval of cathetermediated obstruction of coronary move could lead to important ischaemia and danger of arrhythmia. B) the Judkins proper coronary catheter requires cautious clockwise rotation prior to engagement of the best coronary ostium. B) Injection of contrast throughout angiography leads to extravasation of contrast into an intimal dissection (arrow). Subsequent angiography (this time with an Amplatzer formed catheter) confirmed full healing of the dissection flap. Conal injection and ventricular arrhythmia Special extra care should be taken following intubation of the best coronary ostium. This is often, however not always, heralded by pressure damping, and subsequent injection of distinction can induce ventricular arrhythmia. For this purpose, injection of a small take a look at shot of distinction is really helpful following intubation and exclusion of stress damping to ensure correct positioning. However, provocation testing at angiography with ergonovine or acetylcholine is now rarely carried out. Upon engagement of the Judkins left four diagnostic catheter there was extreme stress damping. Right coronary angiogram earlier than (A) and after (B) injection of intracoronary nitrate. Following location of the catheter within the aortic root, mild clockwise rotation is used to engage the ostium. Catheters for saphenous vein grafts the location of vein graft ostia is highly variable according to surgical approach and aortic root anatomy. Radial catheters the usual catheters utilized in femoral angiography may also be used when the radial route is employed. Fundamentals of fluoroscopy: limitations of angiography It is essential to appreciate that the picture produced throughout coronary angiography is a two-dimensional illustration of a threedimensional structure; i. Overlapping coronary arteries Coronary arteries that lie at completely different depths within the imaging plane will seem as overlapping vessels. During acquisition of an angiographic collection, nice care should be taken to make sure that all overlapping segments have been adequately demonstrated. From left to right; Judkins proper 4, proper coronary bypass catheter, Multipurpose 1 catheter, Sones 1 catheter. An alternative strategy, which assesses the practical effect of an equivocal stenosis, is offered by measurement of intracoronary strain or circulate and the instruments to achieve this are discussed in Chapter 10. Negative remodelling Coronary angiography delineates coronary luminal anatomy and is therefore a wonderful device for the identification of stenotic or occlusive coronary disease. This concern of non-stenotic coronary disease is of specific relevance in acute coronary syndromes where plaque rupture events could occur in apparently undiseased vessels. Bifurcation points Issues of vessel overlap and foreshortening are significantly related at bifurcation points where disease is frequent and simply missed. However, eccentric plaque might only be seen in profile from particular fluoroscopic projections where the encroachment of plaque into the vessel lumen could be absolutely appreciated. This requires imaging during contrast injection into the left and right coronary ostia in a variety of projections. In these views of the proper coronary artery, the discrete stenosis within the posterior descending department (arrowed) is simply clearly demonstrated in the posteroanterior cranial view (C) however is obscured by overlapping vessels in both the left anterior indirect (A) and right anterior oblique (B) projections. Of basic importance is the understanding that the vary of views used and fluoroscopic angles utilized must be adjusted for each affected person to acquire optimal pictures and minimize exposure to distinction and radiation. Positioning of the angiographic picture, magnification, filters, and coning Following engagement of the coronary ostium and previous to acquisition of the angiogram, the radiographer, guided by the operator, will amplify the picture. Further projections shall be required to enable complete assessment of the proximal left anterior descending territory. Adjustment of the relative diploma of angulation is usually required to acquire an optimum picture of this necessary anatomical location. In this posteroanterior cranial projection the relationship between the proximal left anterior descending coronary artery illness and a severe stenosis at the origin of the primary diagonal branch (arrowed) is clearly delineated. Careful modification of ordinary projections is commonly required to reveal bifurcation disease adequately. However, in the right anterior indirect view (B) a discrete eccentric plaque is clear. B) A proper anterior oblique caudal projection exhibits right coronary collateralization (arrows) from the circumflex. The exact projections used for picture acquisition will differ depending on the course of the graft, the native coronary anatomy past its insertion, and the placement of any illness. In panel A the catheter tip is positioned to centrally in this right anterior oblique caudal shot. Filters are wedge-shaped and are positioned to stop picture degradation from areas of higher X-ray penetration. Non-coronary assessment throughout coronary angiography In addition to delineation of the coronary anatomy, further data could additionally be obtained during a coronary angiogram according to the clinical context.
Buy discount diclofenac gel 20 gm on lineTypically in people and most different mammals arthritis in back hereditary cheap diclofenac gel 20 gm with mastercard, the isotonic answer corresponds to zero arthritis valgus knee diclofenac gel 20 gm buy low price. A hypertonic answer accommodates a higher focus of dissolved substances than the red blood cells, which cause the red blood cells to shrink. In contrast, a hypotonic resolution incorporates a decrease focus of dissolved substances than the pink blood cells, inflicting the purple blood cells to swell and presumably burst. Sterility is assured by a three-step process: (1) use of sterile starting supplies and course of gear; (2) use of particular technique in drug product manufacture that minimizes the possibility of contamination from human or extraneous materials throughout Parenteral drug products 543 manufacture; and (3) sterilization postmanufacture, preferably in final marketed sealed containers. The are several strategies of sterilization for parenteral merchandise, including dry heat, steam, filtration, gasoline, and radiation. Filtration is the strategy of choice for solutions that are unstable to other kinds of sterilizing processes, for example, thermolabile products. However, macromolecules, corresponding to proteins and peptides, could additionally be broken by filtration as a outcome of shear stress, leading to alteration of their three-dimensional structure. Therefore, drug interactions with filter supplies are carefully investigated before implementing this methodology of sterilization. However, this methodology requires greater temperature (~160�C�250�C) and longer publicity (~30�180 min) to obtain sterility. A main problem related to dry warmth sterilization is nonuniform distribution of temperature. Steam sterilization is the strategy of alternative for sterilization of aqueous solutions, glassware, and rubber articles. Microorganisms are sometimes killed through the use of -rays, -rays, X-rays, and accelerated electron beams. Thermolabile drugs, corresponding to penicillin, streptomycin, thiamine, 544 Pharmaceutical Dosage Forms and Drug Delivery and riboflavin have been successfully sterilized by ionizing radiation. However, the retail and hospital pharmacists have little opportunity to use radiation sterilization. Systemic drug absorption occurs extra rapidly than from oral administration compared to intravenous administration. Buffers are utilized in parenteral merchandise to stabilize the solution against pH adjustments. During aseptic filtration, the answer is handed by way of a sterile filter of 2 m pore measurement. Heat and radiation sterilization methods are supposed to get rid of viable microorganism from the final merchandise. Their semisolid state and plastic rheological conduct is designed to aid their software to the target surface as a film. Most of the semisolid formulations are used topically to ship drugs to/through the pores and skin. They can be used for topical or systemic drug motion in/through the eye, nostril, ear, vagina, rectum, buccal tissue, or the urethral membrane. In addition, unmedicated semisolid formulations are frequently used as protectants or lubricants. Topical purposes can be designed for both local effects or systemic absorption. For instance, a topical dermatological product is designed to deliver a drug into the pores and skin for treating dermal problems. A transdermal product is designed to ship medication through the skin (percutaneous absorption) to the underlying tissue or the systemic circulation. Topical brokers are used as protectives, adsorbents, emollients, and cleaning agents. An ointment usually incorporates <20% water and different unstable elements, such as ethanol, and >50% hydrocarbons, waxes, or polyols. Ointments are designed to soften or melt at physique temperature, spread simply, and have a easy, nongritty really feel. Ointments are usually used as (1) emollients to make the skin more pliable, (2) protecting limitations to prevent harmful substances from coming in contact with the skin, and (3) automobiles for hydrophobic medication. Ointment bases are classified into 4 basic groups: (1) hydrocarbon bases, (2) absorption bases, (3) emulsion or water-removable bases, and (4) water-soluble bases (Table 23. These bases are used for his or her emollient impact (to hydrate the skin) and as an occlusive dressing. Thus, they are often protective to water labile medication, similar to bacitracin and tetracycline. Oily- or fatty-base ointments may have exhausting, soft, or liquid paraffin bases, or mixtures of these, in such proportions as will render an ointment to be of appropriate consistency. Common hydrocarbon bases embrace the next: � Petrolatum: It is used as a base for water-insoluble ingredients. Yellow petrolatum or petrolatum jelly, for example, Vaseline, melts at 38�C�60�C. Petrolatum varieties an occlusive film on the pores and skin and absorbs less than 5% water underneath normal conditions. These esters embody glycerol monostearate, isopropyl myristate, isopropyl palmitate, butyl stearate, and butyl palmitate. In addition, lanolin derivatives, similar to lanolin oil and hydrogenated lanolin, are typically used. It is beneficial for the extemporaneous preparation of ointments by chilly incorporation of medication, thus being suitable for heat-labile compounds. Anhydrous bases that allow the incorporation of aqueous options, ensuing in the formation of w/o emulsions. These absorption bases are anhydrous autos composed of a hydrocarbon base and an additive. The hydrocarbon base could presumably be, for instance, hydrophilic petrolatum and anhydrous lanolin. The additive is a miscible substance with polar groups (a surfactant), which capabilities as a w/o emulsifier. For instance, ldl cholesterol, lanosterol and different sterols, acetylated sterols, or the partial esters of polyhydric alcohols, such as monostearate or monooleate, can serve as components. Bases that are already w/o emulsions (emulsion bases) and permit the incorporation of small extra portions of aqueous solutions. Lanolin is a w/o emulsion that can type an occlusive movie on the pores and skin and function an emollient, effectively preventing epidermal water loss. Cold cream is a semisolid white w/o emulsion prepared with cetyl ester wax, white wax, mineral oil, sodium borate, and purified water. Sodium borate combines with free fatty acids present in the waxes to form sodium salts of fatty acids (soaps) that act as emulsifiers. For example, Eucerin cream is a w/o emulsion of petrolatum, mineral oil, mineral wax, wool wax, alcohol, and bronopol. It incorporates urea because the active ingredient and is used to help rehydrate dry, scaly skin. The majority of dermatologic drug merchandise are formulated in an emulsion or cream base.
Diclofenac gel 20 gmThe gasoline phase arthritis treatment los angeles diclofenac gel 20 gm discount with amex, on the opposite hand equine arthritis relief buy diclofenac gel 20 gm on-line, is the least ordered, with the molecules undergoing random Brownian motion, impartial of different molecules. The bonds that hold a phase collectively are van der Waals pressure, ionic, dipole, and hydrogen bonds-depending on the atomic structure of the molecules of a section. For instance, water molecules are held collectively predominantly by hydrogen bond and dipole forces, whereas octane molecules are held together by weak van der Waals forces. The strength of intermolecular forces of attraction and the proximity of the molecules comply with the final development: solids > liquids > gases. In the bulk of a part, a molecule is surrounded by different molecules of the same type and encounters similar forces in all instructions, which are inclined to neutralize each other. Forces of attraction between the molecules of the identical kind within a section could be termed cohesive forces, and the resulting phenomenon is termed cohesion. Similarly, forces between the molecules of different sorts at the interface may be termed adhesive forces, and the ensuing phenomenon is termed adhesion. At the liquid�gas interface, cohesive forces are typically greater than adhesive forces, leading to an inward pull on the molecules towards the majority. This pressure pulls and retains the molecules of the interface together and tends to contract the surface, leading to minimization of the uncovered surface area. The molecules on the floor experience enticing forces from all directions, besides on the interface, resulting in a pull toward the majority of the liquid. Surface pressure of the answer forming the movie is a function of the force that should be utilized to break the movie over the size of a movable bar involved with the film. Since the film has two liquid�gas interfaces (one above and one beneath the aircraft of the bar), the whole size of the contact is the same as twice the length of the bar. In different phrases, the amount of force required, or work accomplished, to create further floor is directly proportional to the quantity of new floor being created. While the term floor tension is reserved for liquid�gas and solid�gas interfaces, the time period interfacial rigidity is often used for liquid�liquid interfaces. Interfacial rigidity has the identical symbol and items (dyne/cm) as surface pressure and is derived similarly from the amount of force required to create new interface. Subscripts are commonly used to distinguish between different interfacial tensions. For example, L/L is the interfacial pressure between two liquids (designated "L"), and L/V is the surface tension between a liquid and its vapor (designated "V") within the fuel section. Usually, the interfacial tension (liquid�liquid) of a hydrophilic liquid is lower than its surface rigidity (liquid�vapor). This is because the adhesive forces between two liquid phases forming an interface are generally larger than these between a liquid and a gas section. For example, at ~20�C, the interfacial pressure between water and carbon tetrachloride is forty five mN/m, whereas the surface tension of water is seventy two. These devices measure the pressure by which a floor is held together whereas the drive is utilized on the floor to expand it. The strategies for surface pressure measurement embody the du Nouy technique (maximum pull on a rod or plate immersed in a liquid), du Nouy ring methodology (maximum downward pressure on a hoop pulled by way of the liquid�air interface), Wilhelmy plate technique (downward drive on a plate lowered to the surface of the liquid), and pendant drop technique (shape of the drop at the tip of needle by optical imaging). All of those methods measure the inherent drive inside a liquid that resists the growth or expansion of its surface. Factors affecting this force, or the floor rigidity, of a liquid embrace the following: � Nature of the liquid: Greater the cohesive forces between the molecules of a liquid, higher its surface tension. Interfacial phenomena 207 � Temperature: Surface rigidity of most liquids decreases linearly with an increase in temperature. This is due to larger Brownian movement of individual molecules that results in discount in the intermolecular attractive forces and, thus, the decreased inward pull of the molecules on the surface. Surface free vitality represents the amount of energy put into the system per unit enhance in surface space. Thermodynamically, floor free energy represents the Gibbs free energy at fixed temperature and strain. This phenomenon is thought 208 Pharmaceutical Dosage Forms and Drug Delivery as adsorption. Adsorption refers to the floor binding of a liquid or gas molecule (adsorbate) onto a stable floor (adsorbent). Examples of adsorbents are highly porous solids, corresponding to charcoal and silica gel, and finely divided powders, such as talc. A bodily adsorbed gas could additionally be desorbed from a stable by increasing the temperature and reducing the pressure. Adsorption is a surface phenomenon, distinct from absorption, which implies the penetration via the solid floor into the core of the stable. Since adsorption is a result of an adhesive process, whereby two forms of molecules interact with one another, the character of the 2 kinds of molecules will determine their enticing interactions. Greater the surface space of the adsorbent, more the absolute amount of adsorbate that can be adsorbed. In modeling the adsorption phenomenon, the amount of adsorbate per unit adsorbent is usually calculated. In this state of affairs, the precise surface area (surface area per unit mass) of the adsorbent performs a role in determining the quantity of adsorbate per unit mass of the adsorbent. This phenomenon signifies that a finely divided strong (of the identical mass as a coarse particulate solid) would adsorb larger amount of adsorbate. Temperature increases molecular movement, and its impact on adsorption is determined by the relative change within the intermolecular forces of attraction between the molecules of the 2 phases. Generally, a rise in Brownian motion with rising temperature reduces adsorption. Generally, greater the solute (adsorbate) partial strain or concentration, greater the rate of adsorption. It is usually mediated by weak noncovalent forces of attraction, such as van der Waals Interfacial phenomena Table eight. For example, oxygen adsorbed by carbon is eliminated as carbon dioxide Restricted to formation of monolayer Proceeds at a finite price,which increases quickly with rise in temperature forces, and is reversible. Physical adsorption is an exothermic process, since heat is released with the formation of enticing interactions between molecules of the two phases. Physical adsorption may be related to three phenomena: � Monolayer formation: Adsorption of a solute on a stable surface results in a monolayer formation, because the solute occupies the available surface in a single layer. Once the monolayer formation is full and the situations (such as solute focus within the liquid or partial stress of the gas) are supportive, multimolecular adsorption could happen. These isotherms are characterised by rapid solute adsorption to a limiting worth, which sustains for sure enhance in the partial strain of the solute. An asymptote towards a limiting value is observed after each additional layer formation.
Diclofenac gel 20 gm buy discount lineThe diploma of freedom is calculated primarily based on the sample size and sometimes also the number of variables studied arthritis pain vs bone cancer pain purchase 20 gm diclofenac gel overnight delivery. The degrees of freedom affect the distribution plot of the check statistic and thus the P worth for a given facet joint arthritis in back best diclofenac gel 20 gm. Having calculated the P worth and the check statistic, the given take a look at of significance is carried out per the steps outlined earlier. For instance, if the worth of check statistic obtained for a given take a look at of significance is zero. Each variable or issue may be studied at completely different ranges, indicating the intensity. For example, a clinical research that evaluates one dose of an experimental drug is a one-variable one-level experiment. A examine that evaluates two doses of an experimental drug can be a one-variable two-level research. Another research that evaluates three doses of two experimental medication can be a two-variable three-level examine. The degree may be a quantitative number, such as the dose within the above examples, or it might be a numerical designation of the presence or depth of an effect, such as "0" and "1. Different subgroups of the experimental knowledge points can be subjected to completely different ranges of the treatment, i, the place i = 1, 2, three. If the levels of the therapy are fastened, the model is termed fixed-effects model. On the opposite hand, if the levels of the treatment are randomly assigned from several attainable levels, the mannequin is termed random-effects model. Whether the degrees of a variable or treatment are fastened or random is dependent upon the design of the experiment. A mounted effects model is exemplified by three subgroups of a group of 18 volunteers chosen for a pharmacokinetic study of a given drug at dose levels of zero, 50, and a hundred mg. A random effects model can be exemplified by three subgroups of a gaggle of 18 volunteers chosen for a pharmacokinetic examine of three completely different medication A, B, and C at unknown and variable dose ranges. The choice of a study design as a fixed- or random-effects mannequin is important to the accuracy of knowledge interpretation. The calculation of variance between remedy teams is completely different between fixed- and random-effects model. Although many software program tools are presently out there that cut back the requirement for tedious calculations, it is necessary to perceive the calculations of statistical tests of significance from first principles. Mean of all samples in the experiment is calculated by adding all observations and dividing by the entire variety of samples within the experiment. An F-ratio is computed as the ratio of imply squares of issue impact to the imply sq. of error effect. The F-ratio is in comparison with the P worth for the F-test at designated degrees of freedom to determine the significance of observed results. Hypothetical results of this examine by way of discount of blood ldl cholesterol stage are 132 Pharmaceutical Dosage Forms and Drug Delivery Table 5. In this instance, though the information do look significantly totally different when reviewed with out statistical evaluation, the excessive random error within the observations leads to lack of statistical significance. An alternate means to test the speculation is to use the usual tables to determine the p-value associated with the observed F-value. For example, in the above calculations, the p-value related with the observed F-ratio is zero. For instance, three components evaluated at two completely different levels could be a 23 factorial experiment and two factors evaluated at three completely different levels would be a 32 experiment. An instance of such studies is the effect of temperature and pressure on the progress of a reaction. If the experiment have been run at three ranges of temperature and stress, it might be a 32 factorial experiment, with the total number of experimental runs = 3 � three = 9. For instance, a full-factorial four-factor, two-level research would involve 24 = 2 � 2 � 2 � 2 = sixteen experimental runs. Full-factorial experiments provide data on both the main results of various factors and the results of their interactions. For example, a partial-factorial four-factor, two-level research would contain 24-1 = 16/2 = 8 experimental runs. Partial-factorial experiments present info on the principle results of various components but not on the interaction results. Design and interpretation of partial-factorial experiments are past the scope of this chapter. Hence, the worth of every data point in an experiment is represented by means of the mean of all samples and deviations arising from the impact of two treatments or variables being studied (individual or primary results, i and j and results arising from interplay of these variables, ij) and random variation (ij). The variables in this experiment are commonly termed elements, and the experiment is termed a factorial experiment. Pharmacy math and statistics 137 � No distinction between the population means of samples handled with completely different mixtures of different levels of factors A and B (For example, if each components A and B had two levels - excessive and low - the mixtures might be excessive [A] with low [B] versus low [A] with high [B]. A study of this interaction reveals whether the effect of issue A is totally different when factor B is low versus excessive or not. It is calculated by subtracting the imply value for every column from the imply of all samples, squaring this worth, and including for all columns. Test the speculation: the F-ratio is in comparability with the P value for the F-test at designated degrees of freedom to determine the importance of observed outcomes. This tabular output of outcomes summarizes statistical parameters associated with the data, adopted by a summary of calculated results in a tabular format. The crucial F-value and the p-value related to the calculated F-value are indicated to facilitate hypothesis testing. Thus, on this instance, the contribution 140 Pharmaceutical Dosage Forms and Drug Delivery Table 5. Yield of a chemical synthesis response was studied as a function of temperature and pressure in a 22 full-factorial research without replication. The knowledge, by method of share yield, are summarized in the table Temperature (�C) forty 60 Pressure: 1 atm ninety five. How many milliliters of amoxicillin suspension containing 250 mg/5 mL should be administered to a patient in need of a 400-mg dose of amoxicillin Given your answers to (a) and (b) above, how much sodium would the affected person be taking per day if the affected person is dosed four hundred mg t. Cyclophosphamide is out there as 50-mg tablets and has a really helpful dose of 5 mg/kg o. For a drug with the grownup dose of a hundred mg/kg, what can be the dose for a 4-feet-tall 8-year-old baby weighing 80 lbs Calculate the NaCl equivalents (E value) for the next three medication, given that NaCl has a molecular weight of 58. For the prescription noted below, calculate the NaCl equivalents current within the formulation.
Marsh Citrus (Marsh Tea). Diclofenac Gel. - Are there safety concerns?
- Are there any interactions with medications?
- Pain and swelling of the muscles and joints, whooping cough, bronchitis, colds, cough, stimulating milk flow, increasing sweating, fluid retention, abortion, and other conditions.
- How does Marsh Tea work?
- What is Marsh Tea?
- Dosing considerations for Marsh Tea.
Source: http://www.rxlist.com/script/main/art.asp?articlekey=96156
20 gm diclofenac gel purchase with mastercardTherefore arthritis in border collie dogs diclofenac gel 20 gm on-line, any worth larger than the indicated line has a decrease than 5% chance of occurrence and is said to fail in the area of rejection gouty arthritis feet diclofenac gel 20 gm order on line. This is one-tailed speculation, since knowledge values on only one aspect of the mean are being thought of for speculation testing. Data values that are more extreme than the line are stated to fall in the region of rejection. The p-value is the fractional probability of accepting the null speculation, assuming that the null hypothesis is true. In other phrases, decrease the p-value of the check, expressed as fractional likelihood. The generally used likelihood stage for accepting the null hypothesis is 5%, comparable to the p-value of 0. In different phrases, 124 Pharmaceutical Dosage Forms and Drug Delivery greater the power of the check, expressed in %, larger the prospect that true differences between two totally different sample sets could be detected. The likelihood of type I error is higher when the chosen degree of significance, is greater. For example, the differences between means are examined utilizing t-test, the variations between proportions are examined using z-test, and the differences in the frequency of a categorical variable are tested using 2 take a look at. Commonly used checks of significance, an instance scenario, underlying assumptions of tests, statement of null speculation, and calculations of the test statistic are summarized in Table 5. It must be noted that these checks of significance invariably contain: � the calculation of a check statistic, which represents the difference between the anticipated and the noticed values, or the values of two samples. It also takes into consideration the variability within the pattern through incorporation of normal error. The calculation of take a look at statistic includes quantifying the extent of observed variations vis-�-vis the variability. Pharmacy math and statistics To check difference Tablet friability take a look at was carried out between on a batch on 10 different matched pairs, occasions. Pharmaceutical Dosage Forms and Drug Delivery To check whether a categorical variable follows a hypothesized frequency distribution, use the chi-square goodness-of-fit take a look at. A controlled-release capsule H0: Ps = Ph formulation makes use of drug microspheres H1: Ps Ph encapsulated in onerous gelatin capsules. Three forms of microspheres are encapsulated: 30% w/w of quick launch, 35% w/w of delayed release by 2 h; and 35% w/w of delayed release by four h. Does this pattern represent the targeted quantity for every capsule within the formulation Three results of this drug for every the identical had been tracked-reduction in blood categorical frequency pressure of no less than 20 mm Hg, and variable i distribution in skin rashes and nausea as opposed in each two or more events. Pharmacy math and statistics 127 128 Pharmaceutical Dosage Forms and Drug Delivery � Identifying the likelihood value related to the take a look at statistic at a given degree of significance (P) for the given degrees of freedom. Calculate the amount of NaCl equivalents that would need to be added to the above formulation to make it isotonic for ophthalmic administration. If NaCl have been incompatible with a number of of drugs, how a lot dextrose (molecular weight = 180) could additionally be used instead. Glycerin is a extremely viscous liquid that could be weighed as a substitute of measured in volume. What could be the equivalent weight of calcium chloride (CaCl 2) if its molecular weight is 111 g/mol How many milligrams of this impurity could be ingested by a mean 150-lb adult human being if the drug is to be administered in doses of 5 mg/kg/day in 4 divided doses By reviewing above results, which of the three data sets has the highest unfold around the central tendency By reviewing the above results, which of the three data units has the least unfold around the central tendency By reviewing the above results, the differences between the technique of which two knowledge sets are most probably to be statistically vital By reviewing the above results, the variations between the means of which two data sets are least more likely to be statistically significant Describe the types of molecular forces involved in the formation of coordination and molecular complexes. Describe the influence of plasma�protein binding on the plasma concentration and biodistribution of medication. The resulting structure, by which the ligand is certain to the substrate, known as a complex. In the case of an administered drug binding a physiological protein, the drug is the ligand and the protein is identified as the substrate. A ligand usually has the power to advanced various sorts of substrates with related binding site-in phrases of molecular measurement, geometry, and charge distribution. Similarly, a substrate can bind a number of completely different ligands of comparable dimension, form, and surface properties. In the case of binding of two small-molecule compounds, either of the 2 compounds can be called a ligand or a substrate, relying on the molecular mechanism of interaction. Theophylline and ethylenediamine advanced to yield the bronchodilator drug aminophylline. Thus, on complexation, properties of the drug such as solubility, stability, partitioning (hydrophilicity/lipophilicity), and absorption are altered. Thus, coadministration of a drug that displaces warfarin from its protein-binding websites can cause high free-drug focus, resulting in toxicity of this low therapeutic index drug. Thus, the ionic covalent bonds are formed when an electron-rich atom on the ligand bonds with an electropositive atom of or on the substrate by donating its pair of electrons. Tetracycline complexation with divalent heavy metal cations is an example of a coordination complex. Complexation and protein binding 151 � Molecular complexes: these are noncovalent complexes shaped by a quantity of engaging interactions between two molecules, such as hydrogen bonding, electrostatic attraction, van der Waals forces, and hydrophobic interactions. Their structure involves one or more central steel atom or cation, surrounded by a number of substrates with negatively charged ions (such as carboxylate groups) or impartial molecules possessing lone pair of electrons (such as on nitrogen atoms of amine groups). The number of bonds fashioned between the metal ion and the ligand(s) is recognized as the coordination variety of the advanced. The interplay between the steel ion and the ligand is a Lewis acid� base response, in which the ligand (a base) donates a pair of electrons (:) to the steel ion (an acid) to form the coordinate covalent bond. Several enzymes contain coordination complexation of their amino acids to a number of heavy steel atoms. Coordination complexes play a crucial function in controlling the structure and function of many enzymes. Heavy metal ions present in physiological proteins and enzymes facilitate the formation of coordination complexes that end result within the performance of the protein or the enzyme.
Diclofenac gel 20 gm cheap on lineStarch is carboxymethylated by reacting with sodium chloroacetate in an alkaline medium adopted by neutralization with citric arthritis in dogs generic 20 gm diclofenac gel overnight delivery, or another acid symptoms of arthritis in back of knee 20 gm diclofenac gel purchase with amex. Cross-linking could also be achieved by either bodily strategies or chemical methods by utilizing reagents corresponding to phosphorus oxytrichloride or sodium trimetaphosphate. This is a protracted chain hydrophilic polymer that serves as a binder in moist granulation. Synthetic product � Pyrrolidone is produced by reacting butyrolactone with ammonia. This is adopted by a vinylation reaction by which pyrrolidone and acetylene are reacted beneath pressure. The monomer, vinylpyrrolidone, is then polymerized within the presence of a mixture of catalysts to produce polyvinylpyrrolidone, also referred to as povidone. Povidone, a long chain hydrophilic polymer, can also be a generally used binder in wet granulation. Cross-linking of the hydrophilic polymer generates a swellable but insoluble polymer that serves as a disintegrant. Magnesium stearate is ready both by chemical reaction of aqueous resolution of magnesium chloride with sodium stearate or by the interaction of magnesium oxide, hydroxide, or carbonate with stearic acid at elevated temperatures. Magnesium stearate is a nice hydrophobic powder that serves as a lubricant within the manufacture of stable dosage types. A chemical synthesis process is most well-liked over pure raw materials to assure adequate material purity, availability, and consistency of the method. In the case of amorphous medication, processes such as lyophilization and spray drying are used to convert the medicine into stable types for purification, lengthy shelf life, and ease of handling. The term long range signifies that many molecules could additionally be involved within the intermolecular interactions that outline the mounted association of molecules in a crystalline construction. This smallest fixed association of molecules that repeats throughout the crystal is named a unit cell. In a crystalline materials, the arrangement of molecules with respect to each other is properly outlined and never random. The molecules of an amorphous strong could show intermolecular interactions, however these interactions may not repeat constantly over a number of molecules. Therefore, in an amorphous material, the orientation of molecules with respect to each other is basically random. Crystallization is completed by making a Powders and granules 441 state of supersaturation of the solute in an answer. A supersaturated solution has a solute focus higher than the thermodynamic equilibrium solubility of the solute in the solvent. Supersaturation can result in crystallization via the spontaneous formation or extraneous addition (seeding) of nuclei. Nuclei are the associations of few (10s to 100s) molecules with the same intermolecular spatial preparations that characterize the crystal kind. Supersaturation may be achieved in certainly one of a quantity of ways: � Evaporation of solvent from an answer. For instance, cooling the answer may result in supersaturation if the solute has a constructive warmth of solution (increase in solubility with increase in temperature). For example, addition of a miscible solvent that has lower solubility for the solute may lead to the formation of a cosolvent system with lower total solute solubility than the solute concentration. Crystallization would be expected within the case of supersaturated options, and dissolution of the crystals is expected when the answer focus is lower than the saturation concentration. Intrinsic properties of a molecule along with crystallization conditions determine the possibility of existence of different crystalline (polymorphic) or amorphous types of a molecule. Some different molecules can have several crystalline varieties and can also exist in an amorphous state. At a molecular degree, polymorphs differ in the power and nature of intermolecular interactions. Polymorphs differ in the floor exposure of useful groups of the molecule on completely different faces of a crystal. Accordingly, totally different polymorphic forms of a molecule usually differ in their surface properties such as wettability and interparticle interactions leading to differences in dissolution price, bioavailability, and/or chemical stability. For instance, type of solvent, diploma of supersaturation, pH of resolution, price of cooling, extent of mixing, or the presence of impurities in answer can result in the formation of various crystalline forms of a molecule. Crystalline varieties have decrease free power and are thermodynamically more secure than amorphous forms of a molecule. Different crystalline polymorphic forms usually differ in their thermodynamic stability. When a drug substance exists in several polymorphic types, the higher thermodynamic stability of 1 crystalline form over one other is often attributable to the higher strength of intermolecular interactions and/or closer or dense crystal packing. These variations often replicate in the melting level of various crystalline forms. A metastable (less stable) polymorphic kind tends to rework right into a extra steady polymorphic type on storage. Therefore, identification and characterization of polymorphic forms of a drug substance are carried out during new product growth. In addition, the thermodynamically most stable polymorphic form is normally most well-liked for use in a drug product. For example, a high fee of solvent evaporation from a solution of the solute can lead to the precipitation of solute in an amorphous kind. Spray drying entails atomization of an answer adopted by solvent evaporation in a steady move gaseous section at a temperature greater than the boiling level of the solvent. Large evaporating surface area of small droplets of resolution facilitates the fast price of solvent evaporation. Changes in course of parameters for spray drying, for example, droplet size, solute focus, and rate of solvent evaporation can lead to considerably completely different powder properties, corresponding to size, of the precipitated material. Solvent removal from an answer can be utilized to generate powders that include two or extra solid substances intimately mixed together in a exhausting and fast composition. This course of generates powder particles that have one stable dispersed or dissolved in another solid of higher amount. These methods can be utilized to generate and stabilize amorphous forms of a drug substance. Use of hydrophobic excipients, similar to Eudragits, can produce solid dispersions with gradual or sustained drug launch properties. Characterization of pharmaceutical powders includes analysis and quantification of each particle and bulk properties. The measurement of a cube could be described in terms of the size of its aspect or diagonal. Quantitatively measuring and defining the scale of those particles is usually a problem. Nevertheless, the use of finely divided powders in pharmaceutical unit operations requires a numerical description of particle size, preferably as a single number, to enable comparison of different powder types and likewise of various batches of the same material.
Buy cheap diclofenac gel 20 gm on lineTherefore early arthritis in back cheap 20 gm diclofenac gel with amex, the blending of two or more powders with related particle measurement and shape is the most probably to produce uniform distribution of each material within the combine arthritis pain vs nerve pain diclofenac gel 20 gm visa. Particle measurement expressed as diameter of spheres with equivalent sedimentation rate in the air known as aerodynamic diameter. For instance, for crystal packing structures that lead to the publicity of practical teams on the surface, a polymorphic form with higher particular floor space is more more doubtless to present greater depth of such floor phenomenon than one other polymorphic type with decrease particular floor area. Examples of such crystal surface-dependent physical properties include chemical reactivity or surface adsorption within the stable state and the sticking tendency of a material to the stainless steel processing gear throughout pharmaceutical manufacturing. Notably, the spherical form presents the least surface space per unit quantity or weight of the fabric. For instance, crystal habits of a compound decide the crystal faces exposed to the floor of the solid. Solute�solvent interactions during crystallization determine which faces of a crystal grow quicker than others. In basic, faces of the crystal that interact extra with the solvent grow at a slower pace than the faces which have less interplay with the solvent. Thus, crystal shape is a function of the solvent used during crystallization, and one can produce crystals of different shape having the same crystalline or polymorphic form. For instance, milling of a drug substance leads to smaller, irregular-shaped crystals which might be closer to the spherical geometry. Also, pharmaceutical processes similar to granulation, spheronization, and spray drying can produce bigger particles which are closer to the spherical shape. The quantity of powder on each sieve is weighed and expressed as the size fraction is decrease than sieve opening diameter above and is higher than the one below (on which the powder was retained). Thus, sieve evaluation produces a weight distribution of particles in numerous sieve fractions. A powder sample is dispersed in an insoluble liquid or air and is passed by way of a beam of laser light. The angle of light scattering decreases and the intensity of scattered light will increase with the growing particle measurement. Measuring the depth of scattered mild at a particular angle allows the estimation of measurement of the particle scattering the sunshine. This is an inline technique used to generate real-time information throughout chemical synthesis, such as crystallization, and pharmaceutical processing, similar to granulation. A quick spinning laser beam is focused on the pattern by way of a quartz lens in a conical sample. The laser light that encounters a particle is reflected again to the lens, the place a fiber optic collects the light and passes to a detector that quantifies the intensity. The time period between the incident and the reflected mild, the pace of the rotating lens, and the pace of laser light are used to calculate the size of a particle passing through the focus of the laser mild. Collective plot of chord size of a number of particles 450 Pharmaceutical Dosage Forms and Drug Delivery produces a chord size distribution. Changes within the chord length distribution throughout processing are used as a fingerprint of the process dynamics. However, it offers only a two-dimensional image of a three-dimensional particle. Although this system permits versatility with respect to sample sorts that may be examined, the pattern preparation process can introduce bias into the pattern. Automated picture analysis software is incessantly used when quantitation is desired. The density difference between the dispersed phase and the dispersion medium leads to particle separation. It is more generally used for the standard assessment of colloidal methods, such as suspensions and emulsions, performance assessment of superdisintegrants, similar to croscarmellose sodium, and separation of particles of extremely small dimension from the dispersion medium. The electrical conductance modifications when a particle flows via the aperture, with the change in conductance being proportional to the size of the particle. It is often used for counting biological cells and micro organism, utilizing a coulter counter. A comparability of those techniques with respect to their merits, demerits, vary of particle dimension measured, and principle of operation is provided in Table 19. Increase in particle size of the majority powder is mostly desired to improve its processability, such as move properties. Particle measurement of the powders may be decreased by managed crystallization or milling (also referred to as comminution) of preformed particles. More medium commonly used for assessing stability of dispersed methods � Requires suspension of particles in a Electrozone Changes in electrical zero. Not suitable flowing by way of an for dry powders, sprays, and emulsions aperture as � Nature of particles, such as density and nonconducting porosity affects methodology capability dispersed phase passes through � Rapid and reliable method for complete particle counting � Commonly used for cell count determination in blood samples � Pharmaceutical functions limited to analysis investigations Powders and granules 453 Processing steps to change the dimensions of the particles invariably additionally ends in changes in particle shape. Milling of odd-shaped particles, corresponding to needles, tends to scale back their aspect ratio and to change the form toward spherical dimensions. Granulation is commonly accompanied by shear pressure and consolidation of particles into larger particles, which are inclined to have an irregular form with low side ratios. For example, � Absorption of a drug from a dosage type entails dissolution of the drug substance into the absorption medium. The price of dissolution is proportional to the surface area of the drug substance. Changes in the surface area of the granules or the lubricant can immediately impression the surface coverage and effectiveness of the lubricant. Changes within the floor area of the raw materials can considerably influence the reproducibility of granulation. Particle size is comparatively simpler to measure and evaluate amongst totally different powders. Porosity of the particles refers to airfilled solvent accessible channels inside particles. Thus, porosity contributes to the surface space of the particles with out impacting particle measurement or form. A greater porosity particle of the identical dimension and form as a decrease porosity particle will have higher surface space. The rate of disintegration and drug dissolution from granules is decided by the penetration of the dissolution medium contained in the granules, which is set by the porosity of the granules. It is assumed that the surface space accessible to the penetrating medium is representative of the floor space related to the pharmaceutical purposes of the powder. It is usually expressed as particular surface space, which is the surface area per unit weight of the powder. Adsorption of an inert gasoline (the adsorbate) on a solid floor (the adsorbent) is pushed by the weak van der Waals forces of attraction. The fee and extent of adsorption of the fuel is primarily pushed by the partial stress of the gasoline (P). At isothermal (constant temperature) conditions, Freundlich proposed that the mass of fuel adsorbed (x) per unit mass of adsorbent (m) is given by x = k * P1 n m the place k and n are constants. Langmuir proposed an alternative equation to describe fuel adsorption on the solid surface that relies on the idea of monolayer adsorption.
|

